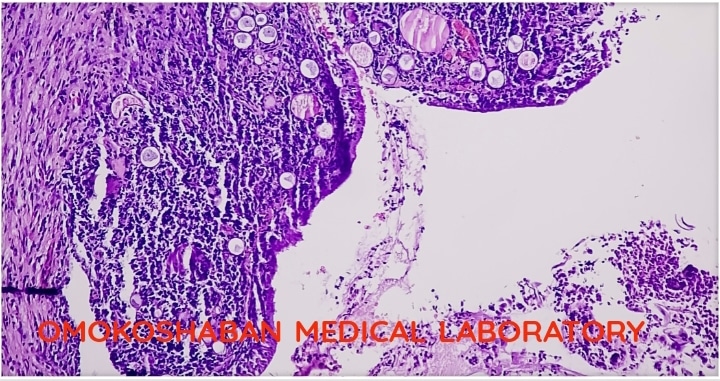
Fibroids in the uterus.
Fibroids, also known as uterine fibroids or leiomyomas, are non-cancerous growths that develop in the muscular wall of the uterus. They can vary in size and number, and may cause symptoms such as heavy or prolonged menstrual bleeding, pelvic pain or pressure, frequent urination, constipation, and infertility.
The exact cause of fibroids is not fully understood, but they are believed to be influenced by hormonal factors, particularly estrogen and progesterone. Fibroids are most commonly diagnosed in women between the ages of 30 and 50, and they are more common in African American women than in women of other racial and ethnic groups.
Treatment options for fibroids depend on the severity of symptoms, the size and location of the fibroids, and the woman’s age and desire for future fertility. Treatment options include medication to control symptoms, such as pain or heavy bleeding, and surgical procedures to remove the fibroids or the entire uterus. In some cases, a minimally invasive procedure called uterine artery embolization can be used to shrink the fibroids by blocking their blood supply.
Causes of Fibroid
The exact cause of fibroids is not fully understood, but they are believed to be influenced by hormonal factors, particularly estrogen and progesterone. Other potential factors that may contribute to the development of fibroids include:
- Genetics: Fibroids tend to run in families, suggesting that there may be a genetic component to their development.
- Hormonal imbalances: Levels of hormones like estrogen and progesterone can affect the growth of fibroids.
- Age: Fibroids are more common in women between the ages of 30 and 50.
- Race: Fibroids are more common in African American women than in women of other racial and ethnic groups.
- Obesity: Women who are overweight or obese are more likely to develop fibroids.
- Diet: Some studies suggest that a diet high in red meat and low in fruits and vegetables may increase the risk of fibroids.
- Environmental factors: Exposure to certain environmental toxins may increase the risk of fibroids, although the evidence is limited.
It’s important to note that while these factors may increase the risk of fibroids, many women who have fibroids do not have any known risk factors.
Symptoms of Fibroid
Fibroids can cause a range of symptoms, depending on their size, number, and location within the uterus. Some women with fibroids may experience no symptoms at all, while others may experience one or more of the following:
- Heavy or prolonged menstrual bleeding: This is the most common symptom of fibroids, and it can lead to anemia or fatigue.
- Pelvic pain or pressure: Fibroids can cause discomfort or a feeling of fullness in the lower abdomen or pelvis.
- Frequent urination: Fibroids that press on the bladder can cause an increased need to urinate, or difficulty emptying the bladder completely.
- Constipation: Fibroids that press on the rectum can cause constipation or difficulty passing stool.
- Back or leg pain: Large fibroids may press on nerves in the lower back or legs, causing pain or numbness.
- Infertility: Fibroids can interfere with fertility if they block the fallopian tubes or distort the shape of the uterus.
It’s important to note that these symptoms are not specific to fibroids and can be caused by other conditions as well. If you are experiencing any of these symptoms, it’s important to speak with your healthcare provider for an accurate diagnosis and appropriate treatment.
Treatment of Fibroid
Treatment options for fibroids depend on several factors, including the size and location of the fibroids, the severity of symptoms, and the woman’s age and desire for future fertility. Treatment options include:
- Watchful waiting: If fibroids are small and not causing any symptoms, your healthcare provider may recommend monitoring them over time and not taking any immediate action.
- Medications: Medications can help relieve symptoms such as heavy bleeding, pain, and pelvic pressure. Examples of medications include nonsteroidal anti-inflammatory drugs (NSAIDs), hormonal birth control, and gonadotropin-releasing hormone (GnRH) agonists.
- Surgery: If fibroids are causing significant symptoms or interfering with fertility, surgery may be recommended. Surgical options include myomectomy (removal of the fibroids while leaving the uterus intact), hysterectomy (removal of the entire uterus), and uterine artery embolization (blocking the blood supply to the fibroids to shrink them).
- MRI-guided focused ultrasound: This is a non-invasive procedure that uses focused ultrasound waves to destroy the fibroids without surgery.
- Endometrial ablation: This is a procedure that removes the lining of the uterus, which can reduce heavy menstrual bleeding caused by fibroids.
The choice of treatment depends on several factors, including the severity of symptoms, the woman’s age, and her desire for future fertility. Your healthcare provider can help you determine which treatment option is best for you.