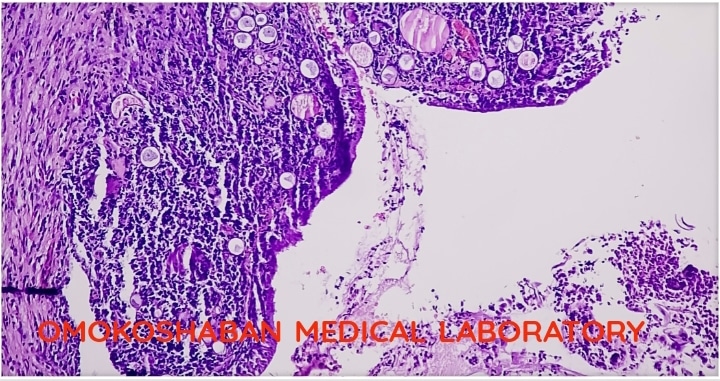
Fibroid in Uterus, Causes, symptoms and treatment
Fibroids in the uterus. Fibroids, also known as uterine fibroids or leiomyomas, are non-cancerous growths…
Malaria Parasites (MP) symptoms, test and treatment
Malaria is a life-threatening disease caused by the Plasmodium parasite, which is transmitted to humans…
Breastfeeding, and it Benefits
Breastfeeding is the process of feeding an infant or young child with milk directly from…
What In-vitro fertilization (IVF)?
In-vitro fertilization (IVF) is a type of assisted reproductive technology (ART) that involves fertilizing an…
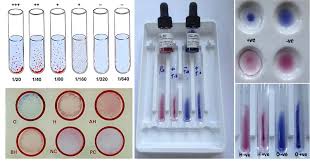
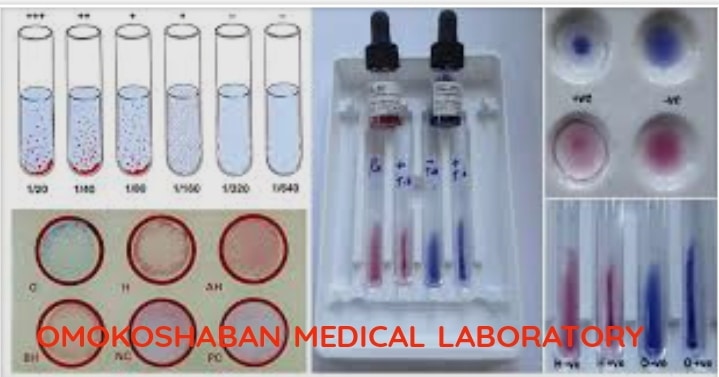
Widal Test Procedure
The Widal test is a serological test used to diagnose typhoid fever, a bacterial infection…
What is Widal Test?
The Widal test is a diagnostic test used to confirm the presence of certain bacteria,…
Fibroid in Uterus, Causes, symptoms and treatment
Fibroids in the uterus. Fibroids, also known as uterine fibroids or leiomyomas, are non-cancerous growths…
Malaria Parasites (MP) symptoms, test and treatment
Malaria is a life-threatening disease caused by the Plasmodium parasite, which is transmitted to humans…
Breastfeeding, and it Benefits
Breastfeeding is the process of feeding an infant or young child with milk directly from…
What In-vitro fertilization (IVF)?
In-vitro fertilization (IVF) is a type of assisted reproductive technology (ART) that involves fertilizing an…
Widal Test Procedure
The Widal test is a serological test used to diagnose typhoid fever, a bacterial infection…
What is Widal Test?
The Widal test is a diagnostic test used to confirm the presence of certain bacteria,…
-
Product on salePetri Dish₦1,700.00
-
Product on saleAutomatic Micro Pipette₦8,500.00
-
Product on saleVis spectrophotometer₦190,000.00
-
Product on saleMalaria Test (MP)₦800.00
-
Product on saleGeneral Test: MP, PCV, Widal, FBC, RVS, Blood Group and more₦17,100.00
-
Product on saleHIV1 and HIV2 Test₦1,500.00
-
Product on saleBlood Group Test₦1,000.00
-
Product on saleMP, Widal, PCV and RBS or FBS₦2,500.00
-
Product on saleFull Blood Count (FBC)₦3,500.00
-
Product on saleSexual Transmitted Disease STD Test₦10,000.00
-
Product on saleMP, Widal, and PCV Test₦2,000.00
-
Product on saleElectronic Compact Scale₦13,500.00
-
Product on salePetri Dish₦1,700.00
-
Product on saleAutomatic Micro Pipette₦8,500.00
-
Product on saleVis spectrophotometer₦190,000.00
-
Product on saleElectronic Compact Scale₦13,500.00
-
Product on saleHospital Medical Screen Folding for Ward Screen /Hospital Bed₦45,000.00
-
Product on saleDual Head Stethoscope₦2,500.00
-
Product on saleDigital Thermometer₦1,200.00
-
Product on saleWeighting Scale₦8,000.00
-
Product on saleAccu-Check Strip₦8,500.00
-
Product on saleAccu Lancet₦2,600.00
-
Sphygmomanometer₦7,000.00
-
Product on saleVDRL Strip₦6,500.00
On Sale
-
Product on salePetri Dish₦1,700.00
-
Product on saleAutomatic Micro Pipette₦8,500.00
-
Product on saleVis spectrophotometer₦190,000.00
-
Product on saleMalaria Test (MP)₦800.00
-
Product on saleGeneral Test: MP, PCV, Widal, FBC, RVS, Blood Group and more₦17,100.00
-
Product on saleHIV1 and HIV2 Test₦1,500.00
-
Product on saleBlood Group Test₦1,000.00
-
Product on saleMP, Widal, PCV and RBS or FBS₦2,500.00
-
Product on saleFull Blood Count (FBC)₦3,500.00
-
Product on saleSexual Transmitted Disease STD Test₦10,000.00
-
Product on saleMP, Widal, and PCV Test₦2,000.00
-
Product on saleElectronic Compact Scale₦13,500.00
Best Sellers
-
Product on saleAccu Lancet₦2,600.00
-
Product on saleAccu-Check Strip₦8,500.00
-
Sphygmomanometer₦7,000.00
-
Product on saleDigital Thermometer₦1,200.00
-
Product on saleDual Head Stethoscope₦2,500.00
-
Product on saleElectronic Compact Scale₦13,500.00
-
Product on saleHospital Medical Screen Folding for Ward Screen /Hospital Bed₦45,000.00
-
Product on saleWeighting Scale₦8,000.00
-
Product on saleVDRL Strip₦6,500.00
-
Product on saleMercury thermometer₦1,200.00
-
Product on saleFull Blood Count (FBC)₦3,500.00
-
Product on salePetri Dish₦1,700.00